Relationship Between Functional Performance, Kinesiophobia with Muscular Strength, Fatigue and Quality of Life in Breast Cancer Survivors Women
Relación entre el Desempeño Funcional, la Kinesiofobia con la Fuerza Muscular, la Fatiga y la Calidad de Vida en Mujeres Sobrevivientes de Cáncer de Mama
https://doi.org/10.32635/2176-9745.RBC.2026v72n1.5385
Vitor Alves Marques1; Wanderson Divino Nilo dos Santos2; Maria Sebastiana Silva3; João Ferreira-Batista Junior4; Claudio Andre Barbosa de Lira5; Leonardo Ribeiro Soares6; Filipe Dinato Lima7; Ricardo Borges Viana8; Naiany Pereira Silva9; Ruffo de Freitas-Junior10; Carlos Alexandre Vieira11
1Universidade Federal de Goiás (UFG), Programa de Pós-Graduação em Ciências da Saúde. Goiânia (GO), Brasil. Universidade Estadual de Goiás (UEG). Quirinópolis (GO), Brasil. E-mail: vitoralvesmarques92@gmail.com. Orcid iD: https://orcid.org/0000-0002-3194-9118
2,3,5,8,11UFG. Goiânia (GO), Brasil. E-mails: wanderson.nilo@gmail.com; mssilva@ufg,br; andre.claudio@gmail.com; vianaricardoborges@hotmail.com; vieiraca11@gmail.com. Orcid iD: https://orcid.org/0000-0002-4378-1041; Orcid iD: https://orcid.org/0000-0001-7265-5872; Orcid iD: https://orcid.org/0000-0001-5749-6877; Orcid iD: https://orcid.org/0000-0001-9200-3185; Orcid iD: https://orcid.org/0000-0002-0083-2910
4Instituto Federal do Sudeste de Minas Gerais, Campus Rio Pomba. Rio Pomba (MG), Brasil. E-mail: joao.ferreira@ifsudestemg.edu.br. Orcid iD: https://orcid.org/0000-0002-7541-8212
6,10UFG, Hospital de Clínicas, Centro Avançado de Diagnóstico do Câncer de Mama (UFG/HC/CORA/Ebserh). Goiânia (GO), Brasil. E-mails: ribeiroufg@gmail.com; ruffojr@terra.com.br. Orcid iD: https://orcid.org/0000-0002-9448-6114; Orcid iD: https://orcid.org/0000-0003-4145-8598
7Centro Universitário de Brasília. Brasília (DF), Brasil. E-mail: filipedinato@hotmail.com. Orcid iD: https://orcid.org/0000-0001-5748-7540
9UFG, Programa de Pós-Graduação em Ciências da Saúde. Goiânia (GO), Brasil. E-mail: naianypereira156@gmail.com. Orcid iD: https://orcid.org/0000-0002-7461-7607
Corresponding author: Vitor Alves Marques. Avenida Brasil, 435 – Conjunto Hélio Leão. Quirinópolis (GO), Brasil. CEP 75860-000. E-mail: vitoralvesmarques92@gmail.com
ABSTRACT
Introduction: Breast cancer is the most prevalent neoplasm among women. Psychobiological aspects and muscle performance are affected during treatment of women cancer survivors. Objective: To correlate functional performance and kinesiophobia with muscle strength, fatigue and quality of life in breast cancer survivors women. Method: Cross-sectional quantitative study with 62 women – 32 breast cancer survivors assigned to the survivors group (SG) (age 52.93 ± 8.95) and 30 apparently healthy women to the control group (CG) (age: 52.16 ± 7.59). Functional performance was evaluated by DASH, kinesiophobia by the Tampa Scale, quality of life by EORTC BR-23, fatigue by FACT B+4, muscle strength by isometric test bilateral and one-repetition maximum test (1RM). Pearson's coefficient measured linear correlation of variables with normal distribution and the Spearman correlation, the relationship between the variables that did not present normal distribution. The significance level was p≤ 0.05. Results: The survivors group (SG) showed worse levels of functional performance (p<0.001), strength (p<0.001), 1RM (p=0.011) and higher levels of kinesiophobia than the control group (CG) (p=0.005). Strength showed a negative correlation with kinesiophobia (r=0.295; p=0.019) and functional performance (r=-0.351; p=0.005). Functional performance showed negative correlation with fatigue (r=-0.435; p=0.0004) and quality of life (r=-0.296; p=0.019). Conclusion: Breast cancer survivors women have low levels of functional performance, muscle strength and higher levels of kinesiophobia. The functional performance was negatively correlated with strength and psychobiological aspects. The kinesiophobia was negatively correlated with strength.
Key words: Breast Neoplasms; Physical Functional Performance; Quality of Life/psychology; Kinesiophobia/psychology.
Introdução: O câncer de mama é o câncer mais prevalente entre as mulheres. As mulheres sobreviventes de câncer são muito afetadas nos aspectos psicobiológicos e no desempenho muscular ao longo do tratamento. Objetivo: Correlacionar o desempenho funcional e a cinesiofobia com a força muscular, fadiga e qualidade de vida em mulheres sobreviventes de câncer de mama. Método: Estudo transversal, quantitativo, com 62 mulheres – 32 no grupo de sobreviventes de câncer de mama (GS) (idade: 52,93 ± 8,95) e 30 aparentemente saudáveis no grupo controle (GC) (idade: 52,16 ± 7,59). O desempenho funcional foi avaliado pelo DASH, a cinesiofobia foi avaliada pela escala de Tampa, a qualidade de vida pelo EORTC BR-23, a fadiga pelo FACT B+4 e a força muscular pelo teste isométrico bilateral e pelo teste 1RM. O coeficiente de Pearson foi utilizado para avaliar a relação entre as variáveis que apresentaram distribuição normal e a correlação de Spearman para avaliar a relação entre as variáveis que não apresentaram distribuição normal. O nível de significância foi de p≤0,05 Resultados: O GS apresentou os piores níveis de desempenho funcional (p<0,001), força (p<0,001), teste 1RM (p=0,011) e maiores níveis de cinesiofobia do que o GC (p=0,005). A força muscular apresentou correlação negativa com cinesiofobia (r=0,295; p=0,019) e o desempenho funcional (r=-0,351; p=0,005). O desempenho funcional apresentou correlação negativa com fadiga (r=-0,435; p=0,0004) e qualidade de vida (r=-0,296; p=0,019). Conclusão: As mulheres sobreviventes de câncer de mama apresentaram baixos níveis de desempenho funcional, força muscular e níveis mais altos de cinesiofobia. O desempenho funcional apresentou correlação negativa com a força muscular e os aspectos psicobiológicos. A cinesiofobia apresentou correlação negativa com a força muscular.
Palavras-chave: Neoplasias da Mama; Desempenho Físico Funcional; Qualidade de Vida/psicologia; Cinesiofobia/psicologia.
RESUMEN
Introducción: El cáncer de mama es el cáncer más prevalente entre las mujeres. Las mujeres sobrevivientes de cáncer se ven significativamente afectadas por aspectos psicobiológicos y el rendimiento muscular a lo largo del tratamiento. Objetivo: Correlacionar el rendimiento funcional y la kinesiofobia con la fuerza muscular, la fatiga y la calidad de vida en mujeres sobrevivientes de cáncer de mama. Método: Sesenta y dos mujeres participaron en el estudio, divididas en un grupo control (GC) (edad: 52,16 ± 7,59) y un grupo de sobrevivientes de cáncer de mama (SC) (edad: 52,93 ± 8,95). El rendimiento funcional se evaluó mediante la escala DASH, la kinesiofobia se evaluó mediante la escala de Tampa, la calidad de vida se evaluó mediante la escala EORTC BR-23, la fatiga se evaluó mediante la escala FACT B+4 y la fuerza muscular se evaluó mediante la prueba isométrica bilateral y la prueba de 1RM. Se utilizó el coeficiente de Pearson para evaluar la relación entre variables distribuidas normalmente y la correlación de Spearman se utilizó para evaluar la relación entre variables distribuidas no normalmente. El nivel de significación fue p≤0,05. Resultados: El grupo SC presentó peores niveles de rendimiento funcional (p<0,001), fuerza (p<0,001), prueba de 1RM (p=0,011) y mayores niveles de kinesiofobia que el GC (p=0,005). La fuerza muscular mostró una correlación negativa con la kinesiofobia (r=0,295; p=0,019) y el rendimiento funcional (r=-0,351; p=0,005). El rendimiento funcional mostró una correlación negativa con la fatiga (r=-0,435; p=0,0004) y la calidad de vida (-0,296; p=0,019). Conclusión: Las mujeres sobrevivientes de cáncer de mama presentaron bajos niveles de rendimiento funcional, fuerza muscular y mayores niveles de kinesiofobia. El rendimiento funcional mostró una correlación negativa con la fuerza muscular y aspectos psicobiológicos. La kinesiofobia mostró una correlación negativa con la fuerza muscular.
Palabras clave: Neoplasias de la Mama; Rendimiento Físico Funcional; Calidad de Vida/psicología; Kinesiofobia/psicología.
INTRODUCTION
Cancer is considered a public health problem, especially in developing countries. It is estimated that in the coming decades more than 20 million new cases will occur in the world. Breast cancer stands out among the main cancer types with the highest incidence and mortality in the world female population1. In 2020, approximately 2 million new cases were detected, accounting for 11.6% of all types of cancer diagnosed2,3 in women. The highest incidence rates were found in Europe and North America and the lowest in East Asia4. In Brazil, for each year of the triennium 2023-2025, 73,610 new cases were estimated, representing an adjusted incidence rate of 41.89 cases per 100,000 women5. In the capital of the state of Goiás, 1,970 new cases of the disease were estimated in 2023, with a rate of 74.5 cases per 100,000 women5.
Cancer treatment affects the quality of life and fatigue of breast cancer survivors women (BCS) with negative impacts on the functionality of the upper limbs. In a study conducted by Lee et al.6, the authors identified that nearly 67% of BCS women had pain in their arms, shoulders, or hands. The factors that contributed to the increase in t-level (FACT-T) and discomfort were high fatigue rates, low levels of quality of life, in addition to the surgical procedure and the effects of radiotherapy due to the presence of lymphedema.
In general, breast cancer treatment is associated with worse quality of life and fear of using the ipsilateral limb to the surgery, also known as kinesiophobia7, an excessive, exhaustive and irrational fear to make movements and practice exercises, because the individuals believe that movement will increase pain or contribute to the recurrence of other injuries8. Studies show that phobic behavior is quite common in patients with breast cancer, holding a direct relationship with kinesiophobia9-11.
On the other hand, good levels of muscle strength seem to contribute positively to these women. Muscle strength has been associated with better indicators of quality of life, lower fatigue, better functional performance, and lower levels of kinesiophobia9,12,13. Strength is related to quality of life, especially in domains involving mobility, personal care, functionality, pain or discomfort, and decreased anxiety and depression. In addition to these domains, strength is also related to the domains of physical function, mental health, and physical aspects12,13.
Similarly, functional performance is also related to good strength levels, minimizing the adverse effects of the treatment12. Muscle strength has been a determining factor to reduce fear of movement, especially those caused by increased kinesiophobia9,14.
Although there is evidence in the literature that breast cancer treatment affects quality of life6, fatigue15, dysfunction of upper limbs13 and kinesiophobia9, it is still unclear how these variables relate to functional performance and muscle strength. Therefore, the aim of this study is to correlate functional performance, kinesiophobia with muscle strength, fatigue and quality of life in breast cancer survivors women.
METHOD
The study was conducted at “Hospital das Clínicas da Universidade Federal de Goiás” (HC/UFG). The inclusion criteria of the SG were being in menopause, not participating in any regular program of resistance exercises in the last six months, submitted to mastectomy or breast quadrantectomy without metastasis and lymphedema self-reported at the first visit. The inclusion criteria of the CG were 40 to 60 years old healthy women (no diabetes or high blood pressure, no history of cancer or regular practice of physical activities in the last six months) and BMI between 20 to 35 kg/m2. Exclusion criteria for the SG were diabetes, hypertension or any cardiovascular disease preventing the practice of physical exercise, medical recommendation to not practice exercises, any articular shoulder, elbow or hand lesion restraining exercises practice. Exclusion criteria for the CG were no prior surgeries, no psychological disorder or neurodivergent individual.
Figure 1 portrays the two study visits. After the questionnaires EORTC BR-23 and FACT B+4 have been applied by two skilled and experienced investigators, the women performed the handgrip strength test (HGS), the 1RM test and the bilateral isometric strength test (BIS). Strength tests (HGS, 1RM and BIS) were repeated after answering the questionnaires.
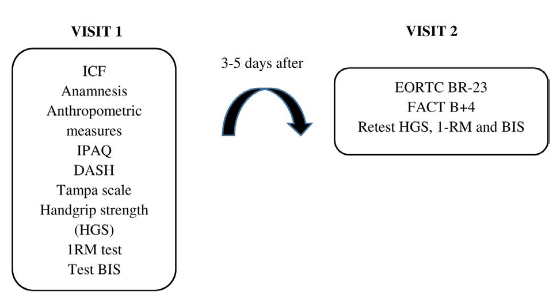
Figure 1. Information collected in the first and second visits
Captions: ICF: Inform Consent Form; IPAQ: International Physical Activity Questionnaire; DASH: Disabilities Arm of Shoulder and Hand; 1RM: one-repetition maximum test; BIS: Bilateral Isometric Strength; EORTC BR-23: European Organization for Research and Treatment of Cancer Quality of Life Questionnaire; FACT B+4: Functional Assessment of Cancer Therapy – Breast Cancer + Arm Scale.
To measure weight and height, the women were barefoot, wearing swimsuits, or comfortable pants and blouses; weight was measured with an analog scale16 (Filizola, mod. Personal 7708, Brazil) and height with stadiometer (Seca, Brazil) according to the procedures described by Gordon et al17. With these two measures, the body mass index (BMI) was calculated.
Physical activity level was evaluated with the 7-question International Physical Activity Questionnaire (IPAQ short version) that estimates the time and duration of a physical activity in different dimensions of activity or physical inactivity18.
The 30-item DASH questionnaire was used to assess the ability of a patient to perform certain upper limb extremity activities, translated, validated and adapted to Brazil by Orfale et al.19. The answers range from zero (no difficulty) to five (inability to perform a task). The DASH score is from zero to 100, obtained by summing the scores of each question and dividing by 1.520.
The Tampa Scale21 of Kinesiophobia is a 17-item assessment checklist to address the fear to perform movements related to physical activity. It uses a 4-point Likert scale (strongly disagree, partially disagree, partially agree and strongly agree). The final score range from 17 to 68. The kinesiophobia classification is divided into negligible (17 to 34 points), intermediate (35 to 50 points) and severe (51 to 68 points).
Isometric tests were applied to measure dynamic muscle strength, (handgrip strength and bench press bilateral isometric) and the one-repetition maximum test (1RM).
The handgrip strength test followed the guidelines of the American Society of Hand Therapists22, consisting in three 3-5s attempts of maximum voluntary contraction, elbow at 90°, with verbal stimulus, alternating right and left and one minute rest interval between attempts22. A digital dynamometer (model EH101, E.clear®) was used.
The bilateral isometric strength (BIS) test was performed on a straight bench press. Elbow flexion was adjusted at ~ 90○ using a manual goniometer (TRIDENT®, Brazil). The warm-up/familiarization comprised three attempts at 50%, 75% and 100% of effort reported by the participant, lasting three seconds per attempt. The rest was one minute between attempts during warm-up. After the warm-up, the participants rested for another three minutes and resumed the test. They performed three to four attempts at maximum voluntary isometric contractions, lasting 5s each23.
Two load cells (S-Type Model OP-312, Optima Scale, US) with a maximum force production capacity of 750 lb were used to acquire data from the bilateral isometric supine test. All digital signal processing was performed by a custom software developed in LabVIEW (National Instruments Corp., Austin, TX, US). This software receives two signals from the designated electronic hardware channel based on the ESP32 microcontroller. The signal, which was sampled at 80 Hz, was transmitted via WiFi in real time and analyzed on a laptop. Before each test day, the system was calibrated using known masses. The force was expressed in Newtons (N) and converted into kilograms of force (kgf).
The dynamic muscle strength measurement was performed through 1RM strength test in the straight supine24. The participants performed three to five attempts of 1RM with three minutes of interval between sets. The warm-up was performed with 50% of the estimated load of 1RM, after warm-up the first attempt was performed with 100% of the estimated load of 1RM, if the participant could perform two repetitions, the loads were adjusted between 5 and 10%.
The quality of life was evaluated by the 23-question European Organization for Research and Treatment of Cancer Quality of Life Questionnaire25 (EORTC - BR 23), a specific instrument to assess quality of life in breast cancer patients. It uses a 4-point Likert scale: 1-not at all, 2-little, 3-moderately and 4-very much. It is calculated separately for each of the scales (symptoms and functional), ranging from zero to 100.
The fatigue was evaluated by the 37-question Functional Assessment of Cancer Therapy – Breast Cancer + Arm Scale (FACT B+4), a multidimensional questionnaire divided into five domains that assesses distinct dimensions of physical well-being (PWB), social and family well-being (SWB), emotional well-being (EWB), functional well-being (FWB) and breast cancer subscale (B). The score FACT TOI – Trial Outcomes Index is identified by combining the subscales PWB, FWB and B. The score is calculated separately for each scale added to each question, ranging from 0 to 164 points26.
The data is presented as standard deviation. Data normality was assessed by the Kolmogorov-Smirnov test. The unpaired test-t was used to compare the groups of variables with normal distribution (handgrip strength, bilateral isometric force and kinesiophobia). The Mann-Whitney test was used to compare the groups of the variables that did not present normal distribution in the 1RM and DASH. The comparison of bilateral isometric force and handgrip strength data between the treated and untreated sides was performed by paired t-test. Pearson's correlation was used to evaluate the relationship between the variables with normal distribution and the Spearman’s correlation, the relationship between the variables that did not present normal distribution. The significance level was considered when p ≤ 0.05.
The Ethics Committee of “Universidade Federal de Goiás” approved the study, report 1,454,981 (CAAE (submission for ethical review: 50717115.4.0000.5083), and of “Hospital das Clínicas da Univerisdade Federal de Goiás” (HC/UFG) (CAAE (submission for ethical review) (Protocol: 50717115.4.3001.5078) in compliance with Directive 466/12 of the National Health Council27.
RESULTS
Table 1 presents the characteristics of the SG and CG.
Table 1. Characteristics of SG and CG
|
Variables |
SG (n=32) |
CG (n=30) |
p |
|
52.93 ± 8.95 |
52.16 ± 7.59 |
0.720 |
|
|
Weight (kg) |
70.04 ± 0.6 |
70.17 ± 12.72 |
0.970 |
|
BMI (kg/m2) |
28.46 ± 4.32 |
28.04 ± 6.95 |
0.950 |
|
Height (m) |
1.57 ± 0.05 |
1.58 ± 0.06 |
0.720 |
|
IPAQ (MET) |
540. 00 ± 68.9 |
606. 00 ± 181.1 |
0.660 |
|
Education (years) |
≥ 8 (16) < 8 (16) |
≥ 8 (23) < 8 (7) |
|
|
Race |
White (16) Non-White (16) |
White (24) Non-White (7) |
|
|
Lymphedema |
Yes (12) No (20) |
- |
|
|
Type of surgery |
Mastectomy (14) Quandrantectomy (17) |
- |
|
|
Type of chemotherapy |
Adjuvant (15) Neoadjuvant (16) |
- |
|
Captions: CG: Control Group; SG: survivors group BMI: Body Mass Index; IPAQ: International Physical Activity Questionnaire; MET: metabolic equivalent.
Table 2. Handgrip Strength, DASH, Kinesiophobia, Fatigue, Quality of Life, Levels of SG and CG
|
Variables |
SG (n=32) |
CG (n=30) |
p |
|
17.05 ± 2.11 |
2.61 ± 6.22 |
< 0.001* |
|
|
Kinesiophobia |
40.50 ± 10.14 |
36.16 ± 6.87 |
0.05* |
|
HGS (max) left (kg) |
25.87 ± 4.73 |
27.99 ± 4.53 |
0.076 |
|
HGS (max) right (kg) |
27.22 ± 4.78 |
28.91 ± 4.66 |
0.165 |
|
BIS (max) left (N) |
106.36 ± 26.89 |
146.65 ± 33.30 |
< 0.001* |
|
BIS (max) right (N) |
109.58 ± 25.57 |
151.93 ± 33.46 |
< 0.001* |
|
BIS (max) total (N) |
214.78 ± 50.29 |
298.58 ± 65.81 |
< 0.001* |
|
1 RM test (kg) |
23.00 ± 4.58 |
28.00 ± 6.01 |
0.011* |
|
FACT G Total |
80.68 ± 12.46 |
88.53 ± 11.56 |
0.013* |
|
FACT B Total |
110.50 ± 16.22 |
124.50 ± 15.96 |
0.002 |
|
General Symptoms |
1.71 ± 0.59 |
1.07 ± 0.36 |
< 0.001* |
|
Side Effect |
1.50 ± 0.72 |
1.143 ± 0.45 |
0.004* |
|
Hair Loss |
1.00 ± 1.29 |
1.00 ± 0.89 |
0.241 |
|
Arm Symptoms |
1.66 ± 0.79 |
1.00 ± 0.36 |
< 0.001* |
|
Breast Symptoms |
1.50 ± 0.63 |
1.00 ± 0.20 |
< 0.001* |
|
Functional Scales |
2.04 ± 0.66 |
1.99 ± 0.62 |
0.744 |
|
Body Image |
1.50 ± 0.78 |
1.00 ± 0.59 |
0.082 |
|
Future Perspectives |
2.00 ± 1.30 |
1.00 ± 0.96 |
0.039* |
|
Sexual Function |
2.00 ± 1.00 |
2.50 ± 1.14 |
0.153 |
|
Sexual Enjoyment |
2.50 ± 1.25 |
2.50 ± 1.35 |
0.260 |
Captions: BIS (max) Left: Left Maximum Bilateral Isometric Strength; BIS (max) Right: Right Maximum Bilateral Isometric Strength; BIS (max) total: Total Maximum Bilateral Isometric Strength; SG: survivors group; CG: control group; DASH: Disabilities of the Arm, Shoulder and Hand; Fact B TOI: Functional Assessment of Cancer Therapy-Breast Total Outcomes Index; Fact G Total: Functional Assessment of Cancer Therapy-General Fatigue Levels; Fact B Total: Functional Assessment of Cancer Therapy Total; HGS: handgrip strength (max) Left; HGS: handgrip strength (max) Right; Kg: kilogram; N: Newton; 1RM: One Repetition Maximum Test; *Value of p<0.05, statistical significance.
Table 3 presents the correlation between the 1RM test, the handgrip strength and the bilateral isometric strength with kinesiophobia and functional performance.
Table 3. Correlation between fatigue, muscle strength and DASH and kinesiophobia
|
Correlated variables |
Kinesiophobia |
DASH |
||
|
|
r |
p value |
r |
p value |
|
- 0.145 |
0.259 |
- 0.480 |
0.000* |
|
|
Social Well-Being |
- 0.005 |
0.682 |
- 0.212 |
0.097 |
|
Emotional Well-Being |
0.002 |
0.851 |
- 0.078 |
0.544 |
|
Functional Well-Being |
- 0.210 |
0.101 |
- 0.343 |
0.006* |
|
Breast Cancer Subescale |
- 0.201 |
0.117 |
- 0.290 |
0.022* |
|
Fact B TOI |
- 0.255 |
0.007 |
-0.465 |
0.0001* |
|
Fact G Total |
- 0.221 |
0.083 |
- 0.435 |
0.0005* |
|
FACT B Total |
- 0.203 |
0.113 |
- 0.346 |
0.0004* |
|
HGS (kg) |
- 0.053 |
0.677 |
- 0.133 |
0.303 |
|
BIS (N) |
- 0.295 |
0.019* |
- 0.351 |
0.005* |
|
1RM test (kg) |
- 0.185 |
0.149 |
-0.833 |
0.519 |
Captions: BIS: Bilateral Isometric Strength; DASH: Disabilities of the Arm, Shoulder and Hand; Fact B TOI: Total Outcomes Index; Fact G Total: General Fatigue Levels; Fact B Total: Fatigue Level Related to Total Breast Cancer; HGS: handgrip strength; *Value of p<0.05, statistical significance; r: Pearson’s correlation coefficient.
The correlation among, DASH and kinesiophobia is presented in Table 4.
Table 4. Correlation among quality of life, DASH and kinesiophobia
|
Correlated variables |
Kinesiophobia |
DASH |
||
|
|
r |
p value |
r |
p value |
|
General Symptoms |
|
|
|
|
|
0.159 |
0.217 |
0.372 |
0.003* |
|
|
Hair Loss |
0.016 |
0.984 |
0.021 |
0.865 |
|
Arm Symptoms |
0.344 |
0.817 |
0.555 |
0.0003* |
|
Breast Symptoms |
0.215 |
0.019* |
0.263 |
0.039* |
|
Functional Scales |
|
|
|
|
|
Body Image |
-0.236 |
0.065 |
-0.296 |
0.019* |
|
Future Perspectives |
-0.420 |
0.645 |
-0.315 |
0.012* |
|
Sexual Function |
-0.224 |
0.482 |
-0.115 |
0.371 |
|
Sexual Enjoyment |
-0.009 |
0.344 |
-0.161 |
0.211 |
Captions: DASH: Disabilities of the Arm, Shoulder and Hand; Value of p<0.05, statistical significance; r: Pearson’s correlation coefficient.
DISCUSSION
The objective of this study was to correlate functional performance and kinesiophobia with handgrip strength, fatigue and quality of life in breast cancer survivors women. Women in the SG have lower BIS levels, 1RM test, levels of fatigue and quality of life. The functional performance was negatively correlated with BIS, fatigue and the functional scales of quality of life, but for general symptoms, the correlation was positive. The kinesiophobia showed a negative relationship with BIS and a positive correlation with general symptoms.
Fatigue negatively affects the SG. The study of Álvarez-Bustos et al.28 identified that approximately 43% of the patients evaluated had fatigue, highlighting that fatigue was related to low levels of muscle strength, obesity, adjuvant chemotherapy, mastectomy, emotional aspects and quality of life. Lovelance et al.29 pointed out that 90% of women diagnosed with cancer have treatment sequelae, fatigue, anxiety and decreased quality of life. Syrowatka et al.30, in a systematic review, concluded that one of the main factors impacting patients’ stress is fatigue, especially related to the physical domain and breast cancer related symptoms. In addition to these factors, the study results show that fatigue has a strong significant relationship in physical well-being, FACT B-TOI and FACT G-Total with the functional performance of the upper limbs. These results show that the physical domains related to fatigue directly influence the lives of breast cancer survivors women.
Zhang and Zhou31 showed that DASH holds a negative mean correlation with fatigue levels (r=-0.564; p<0.005), consistent with the present study. DASH presented an inverse correlation with fatigue levels (r=-0.435; p=0.0005), especially in the domains related to physical well-being (r=-0.480; p=0.0000) and the scales of breast cancer related symptoms (r=-0.345; p=0.0004)32.
In addition to fatigue, quality of life is also influenced during and after treatment of breast cancer patients. Lopes et al.33 analyzed the quality of life of survivors women and identified poor scores of specific domains of quality of life (side effects, breast symptoms, arm symptoms). Moshina et al.34 conducted a systematic review on the factors that affect mental and emotional aspects throughout treatment. The present study showed that the domain of quality of life related to arm symptoms has a direct relationship with the functional performance of the upper limbs, which demonstrates that the post-surgery influence of the affected limb directly interferes with the lives of these women.
The quality of life is negatively influenced by decline of functional performance. Boquiren et al.35 showed that there is a significant correlation (r=0.675; p<0.005) between the high levels of DASH and the low quality of life of women who survived breast cancer. These results are consistent with the present study in regard to side effects (r=0.372, p=0.003) and the arm symptoms (r=0.555, p=0.000). One of the factors that explains this relationship between low levels of functional performance and low levels of quality of life is the limitation of the upper limbs, especially in the shoulders and arms, as they are directly related with symptoms in the breast and arm36.
Corroborating the results above, Altas and Dermidal37 analyzed 70 survivors women post-mastectomy to evaluate the correlation between kinesiophobia and fear, showing that these women hold high positive correlation with fear (r=0.904; p<0.001).
Van der Gucht et al.9 evaluated kinesiophobia levels in breast cancer survivors women throughout treatment. The results show that kinesiophobia levels increase mainly during hormone therapy (p<0.001). Elshahat et al.10 conducted a systematic review of the reasons why these women do not practice physical exercise. Among the reasons presented are kinesiophobia, fatigue, and quality of life. Can et al.10 attempted to correlate functional capacity with kinesiophobia. The results show that kinesiophobia directly influences the decrease in functional performance9, increasing the sensation of pain and discomfort, especially arm, hand and shoulder37, a common problem of cancer survivors impacting the daily activities and reducing the practice of physical activity.
The study limitations are the small sample, it was not analyzed whether lymphedema may influence functional performance and kinesiophobia and whether physical activity may impact these two variables due to potential postoperative complications since there was no stratification according to axillary approach. In addition, the impact of mastectomy or quadrantectomy and the axillary approach on functional performance, kinesiophobia with muscle strength were not investigated separately. No details of the adjuvant and neoadjuvant therapies of each patient were available, which could impact the results. The ideal sample size was not calculated as well.
Future studies should analyze the influence of the type of surgery and treatment on functional performance and kinesiophobia. It is paramount that health professionals are aware to what extent quality of life and fatigue are correlated with kinesiophobia and functional performance as pointed out herein. This knowledge is relevant for better understanding of how breast cancer survivors women are affected psychologically.
CONCLUSION
Breast cancer survivors women have low level of strength and low level of psychobiological aspects than healthy women. In addition, functional performance and kinesiophobia present reverse correlation with strength and fatigue and a direct correlation with the overall symptoms of the disease.
CONTRIBUTIONS
All the authors contributed to the study design, acquisition, analysis and interpretation of the data, writing, and critical review. They approved the final version for publication.
STATEMENT OF DATA AVAILABILITY
All content underlying the text is contained in the manuscript.
DECLARATION OF CONFLICT OF INTERESTS
There is no conflict of interest to declare.
FUNDING SOURCES
None.
REFERENCES
1. Bray F, Laversanne M, Sung H, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 24;74(3):229-63. doi: https://doi.org/10.3322/caac21834
2. Popovic V, Milosavljevic N, Radojevic M, et al. Analysis of postoperative radiotherapy effects within risk groups in patients with FIGO I, II, and III endometrial cancer. Indian J of Can. 2019;56(4):341. doi: https://doi.org/10.4103/ijc.IJC_370_18
3. Wilke L, Andratschke N, Blanck O, et al. ICRU report 91 on prescribing, recording, and reporting of stereotactic treatments with small photon beams: statement from the DEGRO/DGMP working group stereotactic radiotherapy and radiosurgery. Strah. Onkol. 2019;195:193-8. doi: https://doi.org/10.1007/s00066-018-1416-x
4. Global Cancer Observatory [Internet]. Geneva: GLOBOCAN; 2020 [acesso 2025 nov 25]. Disponível em: https://gco.iarc.who.int/today/en/dataviz/bars?mode=cancer&group_populations=1
5. Instituto Nacional de Câncer. Estimativa 2023: incidência de câncer no Brasil. Rio de Janeiro: Instituto Nacional do Câncer; 2022.
6. Lee TS, Kilbreath SL, Refshauge KM, et al. Prognosis of the upper limb following surgery and radiation for breast cancer. Breast Can Res and Treat. 2008; 110(1):19-37. doi: https://doi.org/10.1007/s10549-007-9710-9
7. Saulicz M, Saulicz E, Knapik A, et al. Impact of physical activity and fitness on the level of kinesiophobia in women of perimenopausal age. Przeg Menop. 2016;15(2):104-11. doi: https://doi.org/10.5114/pm.2016.61193
8. Mascarenhas KCS. Caracterização do grau de cinesiofobia em indivíduos com dor lombar crônica inespecífica [monografia]. [Brasília, DF]: Universidade Federal de Brasília; 2018.
9. Van der Gucht E, Dams L, Meeus M, et al. Kinesiophobia contributes to pain-related disability in breast cancer survivors: a cross-sectional study. Support Car in Can. 2020;28(9):4501-8. doi: https://doi.org/10.1007/s00520-020-05304-4
10. Can AG, Can SS, Ekşioğlu E, et al. Is kinesiophobia associated with lymphedema, upper extremity function, and psychological morbidity in breast cancer survivors? Turk J of Phys Med and Rehab. 2019;65(2):139-46. doi: https://doi.org/10.5606/tftrd.2019.2585
11. Elshahat S, Treanor C, Donnelly M. Factors influencing physical activity participation among people living with or beyond cancer: a systematic scoping review. BioMed Central. 2021;18:1-20. doi: https://doi.org/10.1186/s12966-021-01116-9
12. Cantarero-Villanueva I, Fernández-Lao C, Díaz-Rodríguez L, et al. The piper fatigue scale-revised: Translation and psychometric evaluation in Spanish-speaking breast cancer survivors. Qual of Life Resear. 2014;23(1):271-6. doi: https://doi.org/10.1097/PHM.0b013e31825f1538
13. Zabit F, Iyigun G. A comparison of physical characteristics, functions and quality of life between breast cancer survivor women who had a mastectomy and healthy women. J of Back and Musc Rehabilitation. 2019;32(6):937-45. doi: https://doi.org/10.3233/BMR-181362
14. Smoot B, Paul SM, Aouizerat BE, et al. Predictors of altered upper extremity function during the first year after breast cancer treatment. Am J of Phys Med and Rehab. 2016;95(9):639-55. doi: https://doi.org/10.1097/PHM.0000000000000455
15. Fisher MI, Capilouto G, Malone T, et al. Comparison of upper extremity function in women with and women without a history of breast cancer. Phys Ther. 2020;100(3):500-8. doi: https://doi.org/10.1093/ptj/pzaa015
16. Coswig VS, Neves AHD, del Vechio FB. Características Físicas e desempenho motor no jiu-jitsu brasileiro: estudo com iniciantes e experientes na modalidade. Lect Educ Fis y Despor Rev Dig. 2011;16(162):1-9.
17. Gordon CC, Chumlea WC, Roche AF. Stature, recumbent lenght, and weight. In: Lohman TG, Roche AF, Martorell R, editors. Anthropometric standardization reference manual. Champaign: Hum Kin Books; 1988. p. 3-8.
18. Matsudo S, Araujo T, Andrade D, et al. International physical activity questionnaire (IPAQ): study of validity and reliability in Brazil. Rev Bra de Ativ Fís e Saúde. 2001;6(2):5-18.
19. Orfale AG, Araújo PMP, Ferraz MB, et al. Translation into Brazilian Portuguese, cultural adaptation, and evaluation of the reliability of the disabilities of the arm, shoulder, and hand questionnaire. brazilian dash questionnaire brazilian. J of Med and Biol Res. 2005;38(2):293-302. doi: https://doi.org/10.1590/s0100-879x2005000200018
20. Cheng HMS. Disabilities of the arm, shoulder, and hand-dash: análise da estrutura fatorial da versão adaptada para o português [dissertação]. [Belo Horizonte]: Universidade Federal de Minas Gerais; 2006.
21. Trocoli TO, Botelho RV. Prevalência de ansiedade, depressão e cinesiofobia em pacientes com lombalgia e sua associação com os sintomas da lombalgia. Rev Bra de Reumatol. 2016;56(4):330-6. doi: http://dx.doi.org/10.1016/j.rbr.2015.09.009
22. Rogers BH, Brown JC, Gater DR, et al. Association between maximal bench press strength and isometric handgrip strength among breast cancer survivors. Arch of Phy Med and Rehab. 2017;98(2):264-9. doi: http://dx.doi.org/10.1016/j.apmr.2016.07.017
23. Hagstrom AD, Marshall PWM, Lonsdale C, et al. Resistance training improves fatigue and quality of life in previously sedentary breast cancer survivors: a randomised controlled trial. Eur J of Can Care. 2015;784-94. doi: https://doi.org/10.1111/ecc.12422
24. Santos WDN, Vieira A, Lira CAB, et al. Once a week resistance training improves muscular strength in breast cancer survivors: a randomized controlled trial. Integr Can Therap. 2019; 18:1-9. doi: https://doi.org/10.1177/1534735419879748
25. European Organisation For Research and Treatment of Cancer. EORTC QLQ-BR23 Scoring Manual [Internet]. Bruxelas: EORTC; 2008 [acesso 2025 nov 9]. p. 1-32. Disponível em: https://www.eortc.be/qol/ScoringInstructions/BR23%20Summary.pdf
26. Michels FAS, Latorre MRDO, Maciel MS. Validity, reliability and understanding of the EORTC-C30 and EORTC-BR23, quality of life questionnaires specific for breast cancer. Rev Bras Epidemiol. 2013;16(2):352-63. doi: https://doi.org/10.1590/s1415-790x2013000200011
27. Conselho Nacional de Saúde (BR). Resolução n° 466, de 12 de dezembro de 2012. Aprova as diretrizes e normas regulamentadoras de pesquisas envolvendo seres humanos. Diário Oficial da União, Brasília, DF. 2013 jun 13; Seção I:59.
28. Álvarez-Bustos A, Pedro CG, Romero-Elías M, et al. Prevalence and correlates of cancer-related fatigue in breast cancer survivors. Supp Car in Can. 2021;29(11):6523-34. doi: https://doi.org/10.1007/s00520-021-06218-5
29. Lovelace DL, McDaniel LR, Golden D. Long-Term Effects of Breast Cancer Surgery, Treatment, and Survivor Care. J of Mid and Wom’s Health. 2019.64(6):713-24. doi: https://doi.org/10.1111/jmwh.13012
30. Syrowatka A, Motulsky A, Kurteva S, et al. Predictors of distress in female breast cancer survivors: a systematic review. Bres Can Res Treat. 2017;165(1):229-45. doi: https://doi.org/10.1007/s10549-017-4290-9
31. Zhang H, Duan Y, Zhou F. Explore the application value of prospective monitoring model in the nursing management of breast cancer patients during perioperative period. Front in Surg. 2022;23(9):1-19. doi: https://doi.org/10.3389/fsurg.2022.850662
32. Lopes JV, Bergerot CD, Barbosa LR, et al. Impact of breast cancer and quality of life of women survivors. Rev Bras Enferm. 2018;71(6):2916-21. doi: http://dx.doi.org/10.1590/0034-7167-2018-0081
33. Siqueira TC, Frágoas SP, Pelegrini A, et al. Factors associated with upper limb dysfunction in breast cancer survivors. Supportive Care in Cancer. 2021;29(4):1933-40. doi: https://doi.org/10.1007/s00520-020-05668-7
34. Moshina N, Falk RS, Hofvind S. Long-term quality of life among breast cancer survivors eligible for screening at diagnosis: a systematic review and meta-analysis. Publ Health. 2021;1999(1):65-76. doi: https://doi.org/10.1016/j.puhe.2021.08.008
35. Boquiren VM, Hack TF, Thomas RL, et al. A longitudinal analysis of chronic arm morbidity following breast cancer surgery. Breas Can Res and Treat. 2016;157(3):413-25. doi: https://doi.org/10.1007/s10549-016-3834-8
36. Thomas-MacLean RL, Hack T, Kwan W, et al. Arm morbidity and disability after breast cancer: New directions for care. Oncol Nurs For. 2008;35(1):65-71. doi: https://doi.org/10.1188/08.onf.65-71
37. Altas EU, Demirdal ÜS. The effects of post-mastectomy lymphedema on balance, Kinesiophobia and fear of falling. J of Com Heal Nurs. 2021;38(2):130-8. doi: https://doi.org/10.1080/07370016.2021.1887564
Recebido em 28/7/2025
Aprovado em 4/10/2025
Scientific-editor: Anke Bergmann. Orcid iD: https://orcid.org/0000-0002-1972-8777
![]()
Este é um artigo publicado em acesso aberto (Open Access) sob a licença Creative Commons Attribution, que permite uso, distribuição e reprodução em qualquer meio, sem restrições, desde que o trabalho original seja corretamente citado.