ORIGINAL ARTICLE
Expression of BRCA1 and BRCA2 Genes in Patients Undergoing Transrectal Prostate Biopsy
Expressão dos Genes BRCA1 e BRCA2 em Pacientes Submetidos à Biópsia Transretal de Próstata
Expresión de los genes BRCA1 y BRCA2 en Pacientes Sometidos a Biopsia Transrectal de Próstata
https://doi.org/10.32635/2176-9745.RBC.2026v72n1.5460
Eduardo Maciel Narvaes1; Alyna Araújo e Marcondes2; Roberto de Andrade Garcia Filho3; Paulo Gabriel da Silva Mota4; Patrícia de Lima Lemos5; Claudinéia de Araújo6
1-6Universidade Federal de Rondonópolis (UFR), Faculdade de Ciências da Saúde. Rondonópolis (MT), Brasil.
1E-mail: edumuarama@yahoo.com.br. Orcid iD: https://orcid.org/0009-0002-0950-4734
2E-mail: alynaam@gmail.com. Orcid iD: https://orcid.org/0000-0001-7930-9673
3E-mail: robertogarciafilho@hotmail.com. Orcid iD: https://orcid.org/0000-0001-9403-2457
4E-mail: paulogabrieldasilvamota@gmail.com. Orcid iD: https://orcid.org/0000-0003-1614-7907
5E-mail: patricia.lima@ufr.edu.br. Orcid iD: https://orcid.org/0000-0002-5956-4471
6E-mail: claudiaadna@gmail.com. Orcid iD: https://orcid.org/0000-0002-3043-2731
Corresponding author: Eduardo Maciel Narvaes. Avenida dos Estudantes, 5055 – Cidade Universitária. Rondonópolis (MT), Brasil. CEP 78735-901. E-mail: edumuarama@yahoo.com.br
ABSTRACT
Introduction: Prostate cancer (PCa) is the second most common type of cancer among the male population. Men carrying mutations in the BRCA1 and BRCA2 genes are known to have a greater risk of developing PCa. Objective: To correlate clinical and anatomopathological aspects with the expression of the BRCA1 and BRCA2 genes in patients who underwent transrectal prostate biopsy. Method: Transrectal prostate biopsy fragments were collected from 89 individuals. Samples from 84 patients were subjected to RT‒qPCR to evaluate the expression of BRCA1 and BRCA2 transcripts. Results: Of the 84 selected patients, 26 (30.90%) tested positive for PCa and had a PSA level > 10 ng/ml (p=0.019). The median expression of the BRCA1 and BRCA2 genes in negative fragments was greater in PCa-positive patients (p=0.002 and p=0.038, respectively). BRCA1 gene expression was greater in PCa-negative patients than in patients with PCa (p=0.003). There was no statistically significant difference in the expression of the BRCA1 and BRCA2 genes according to Gleason classification or PSA values. Conclusion: The results demonstrated that the median expression of the BRCA1 and BRCA2 genes was greater in negative fragments from patients with prostate cancer than in fragments from individuals without prostate cancer.
Key words: Prostatic Neoplasms/pathology; Genes, BRCA1; Genes, BRCA2; Gene Expression; Neoplasm Grading/statistics & numerical data.
RESUMO
Introdução: O câncer de próstata (CaP) é o segundo tipo de câncer mais comum entre a população masculina. Homens portadores de mutações nos genes BRCA1 e BRCA2 apresentam maior risco de desenvolver CaP. Objetivo: Correlacionar aspectos clínicos e anatomopatológicos com a expressão dos genes BRCA1 e BRCA2 em pacientes submetidos à biópsia transretal de próstata. Método: Fragmentos de biópsia transretal de próstata foram coletados de 89 indivíduos. Amostras de 84 pacientes foram submetidas à RT-qPCR para avaliar a expressão dos transcritos BRCA1 e BRCA2. Resultados: Dos 84 pacientes selecionados, 26 (30,90%) apresentaram resultado positivo para CaP e PSA > 10 ng/ml (p=0,019). A mediana de expressão dos genes BRCA1 e BRCA2 em fragmentos negativos foi maior em pacientes CaP positivos (p=0,002 e p=0,038, respectivamente). A expressão do gene BRCA1 foi maior em pacientes com CaP negativo do que em pacientes com CaP (p=0,003). Não houve diferença estatística significante na expressão dos genes BRCA1 e BRCA2, de acordo com a classificação de Gleason ou os valores de PSA. Conclusão: Os resultados demonstraram que a mediana da expressão dos genes BRCA1 e BRCA2 foi maior em fragmentos negativos de pacientes com câncer de próstata, quando comparados com fragmentos de indivíduos não portadores de neoplasia prostática.
Palavras-chave: Neoplasias da próstata/patologia; Genes BRCA1; Genes BRCA2; Expressão Gênica; Gradação de Tumores/estatística & dados numéricos.
RESUMEN
Introducción: El cáncer de próstata (CaP) es el segundo tipo de cáncer más común entre la población masculina. Los hombres portadores de mutaciones en los genes BRCA1 y BRCA2 tienen un mayor riesgo de desarrollar CaP. Objetivo: Correlacionar aspectos clínicos y anatomopatológicos con la expresión de los genes BRCA1 y BRCA2 en pacientes sometidos a biopsia de próstata transrectal. Método: Se recogieron muestras de biopsia de próstata transrectal de 89 individuos. Se sometieron muestras de 84 pacientes a RT-qPCR para evaluar la expresión de las transcripciones BRCA1 y BRCA2. Resultados: De los 84 pacientes seleccionados, 26 (30,90%) presentaron resultado positivo para cáncer de próstata (CaP) y PSA > 10 ng/ml (p=0,019). La mediana de la expresión de los genes BRCA1 y BRCA2 en los fragmentos negativos fue mayor en los pacientes CaP positivos (p=0,002 y p=0,038, respectivamente). La expresión del gen BRCA1 fue mayor en los pacientes CaP negativos que en los pacientes CaP negativos (p=0,003). No hubo diferencia estadísticamente significativa en la expresión de los genes BRCA1 y BRCA2, según la clasificación de Gleason o los valores de PSA. Conclusión: Los resultados demostraron que la mediana de la expresión de los genes BRCA1 y BRCA2 fue mayor en los fragmentos negativos de pacientes con cáncer de próstata, en comparación con los fragmentos de individuos sin neoplasia de próstata.
Palabras clave: Neoplasias de la Próstata/patologia; Genes BRCA1; Genes BRCA2; Expresión Génica; Clasificación del Tumor/estadística & datos numéricos.
INTRODUCTION
Prostate cancer (PCa) is the second most common type of cancer among the male population. In Brazil, for each year of the triennium 2023-2025, more than 71 thousand new cases are expected, corresponding to an estimated risk of 67.86 new cases per 100 thousand men1. Risk factors include family history of the disease, age, lifestyle, hormonal and genetic aspects. Men of African descent have higher incidence rates, more aggressive forms, and higher mortality rates2.
It is estimated that more than one million new cases were diagnosed worldwide in 2020, making it the tumor with the highest incidence among men in Africa and Latin America3. In Central and South America, the number of cases of prostate cancer is expected to almost double by the year 2030 due to population growth and aging, in addition to increased early detection and greater public awareness4.
In cases of PCa, the anatomical pathology report should provide the histological grading of the Gleason system whose objective is to determine the aggressiveness, probable rate of tumor growth, and tendency to spread, in addition to helping in the determination of the best treatment for the patient. However, the Gleason score is not reliable enough to stratify patients for the presence of pathogenic variants5.
It is known that the hereditary risk of prostate cancer is associated with more aggressive disease. In this context, deleterious mutations of some genes, such as BRCA1 and BRCA2, which were linked to breast and ovarian cancer, have been associated with a high risk of development and increased genetic susceptibility to prostate cancer6,7.
The expression of the BRCA1 gene is a characteristic of more proliferative and aneuploid prostate tumors and is more common in metastatic disease8. Similarly, the expression of BRCA1-associated protein 1 is related to lymphovascular invasion, PSA values greater than 10 ng/ml, and T3b tumors9.
BRCA2 mutations may be associated with a risk of more aggressive, high-grade prostate cancer with a Gleason > 8. In addition, BRCA2 germline infections increase the risk of PCa by 8.6 times in men aged ≤ 65 years10,11.
Given that the early detection of more aggressive forms of this neoplasm is a complex challenge that has not yet been sufficiently explored, the purpose of this study was to determine the expression of the BRCA1 and BRCA2 genes in patients undergoing transrectal prostate biopsy in men treated in Mato Grosso southern region and to correlate it with prostate cancer severity and anatomical pathological characteristics of the fragments analyzed.
METHOD
This was an exploratory, cross-sectional observational study. The study included patients from the regional health consortium in the southern region of the state of Mato Grosso who were selected between June 2019 and October 2020 and asked to undergo prostate biopsy at a urologic service. The sample consisted of all patients who underwent the first transrectal prostate biopsy with clinical indication after the informed consent was obtained. The following exclusion criteria were established: presence of active urinary tract infection and inability to understand or sign the informed consent form.
The study clinical data were obtained from the patients' medical charts, while the anatomopathological data were identified in reports issued by a pathology laboratory accredited to the state health network. Immunohistochemistry analysis, required for inconclusive cases, was performed as a routine diagnostic procedure by this service. Patient classification for this study was based on the final reports issued, and the technical details of the immunohistochemistry protocol were not made available to the research team. The following variables were evaluated: age, PSA value, anatomopathological data, Gleason histological grade and ISUP (International Society of Urological Pathology) grade.
The patients underwent transrectal biopsy by sextants with removal of at least 18 fragments, 12 of whom underwent histopathological analysis and six of them underwent molecular analysis. The procedures were performed under sedation and local anesthesia. The tissue fragments obtained were stored in cryovials preserved in liquid nitrogen at -196°C.
Prostate tissue samples were thawed, weighed, placed in TRIzol solution and homogenized using a Turrax N10 apparatus. After treatment with chloroform, the total RNA was precipitated with isopropyl alcohol. To eliminate DNA contaminants, DNAse treatment was performed for 15 minutes at room temperature, followed by another 10 minutes at 65°C.
For mRNA amplification, RNA (0.5-1 μg) was reverse-transcribed using random primers and SuperScript II RNase H transcriptase according to the manufacturer’s instructions (Invitrogen).
qPCR was performed using a Power SYBR Green Master Mix Kit (Promega) to determine the relative expression levels of the BRCA1 and BRCA2 transcripts.
qPCR was performed with 4 ng/μl cDNA in a final volume of 10 μl in a 96-well plate. All cDNA samples were previously quantified. The GAPDH gene was used as a normalizer for the quantification of the expression of the genes of interest. The basic conditions of the thermocycling reaction were 50°C for 2 minutes, 95°C for 10 minutes, and 40 cycles of 95°C for 15 seconds, 50°C for 30 seconds, and 60°C for 60 seconds12.
Gene expression data were generated using a CFX96 thermal cycler from Bio-Rad, and relative gene expression was determined using ∆∆CT13. For the calculation of ∆∆CT, the group of samples from patients with a negative result for prostate cancer (PCa negative) was used as the reference group (calibrator).
The data were collected and entered into Windows Excel spreadsheets and then transferred to the statistical program Stata14 version 16.0 (StataCorp LP, College Station, United States) for analysis.
For data analysis, Fisher's chi-square test was performed to verify the associations among the categorized variables, age, PSA, and ISUP between the groups. The mean and median variables of age and PSA were also calculated, as well as the frequency of the variables city of origin and occupation.
To verify differences in the medians of gene expression between the groups, the Mann‒Whitney test was performed due to the absence of normally distributed data. The correlation between the variables PSA, BRCA1, BRCA2, and ISUP was verified by Spearman's test, p-value <0.05 indicated statistical significance.
This study was approved by the Ethics Committee of “Hospital Universitário Julio Müller” of “Universidade Federal de Mato Grosso” (HUJM/UFMT) report number 1543035/2016 – CAAE (submission for ethical review): 54636016.1.0000.5541. All the study patients signed the Informed Consent Form (ICF), and authorized the use of biological material for molecular analyses. The study complied with the Declaration of Helsinki and Directives 466/201215 and 510/201616 of the National Health Council.
RESULTS
A total of 89 patients who underwent transrectal prostate biopsy were enrolled in the study. To determine the clinical suspicion of prostate cancer, all samples were sent for histopathological analysis. Among these patients, 20% (n=18) were confirmed to have usual prostate adenocarcinoma, and 19% (n=17) had inconclusive results, requiring immunohistochemical analysis. Therefore, 61% of the biopsies performed had negative histopathological results for prostate cancer.
For immunohistochemical analysis, five of the 17 patients with inconclusive pathological results were excluded. One of them died before the release of the results, and four due to missing data in the medical charts. Of the 12 patients evaluated, eight tested positive for usual prostate adenocarcinoma. Therefore, after pathological and immunohistochemical results were evaluated, 30.90% (n=26) of the patients have been diagnosed with PCa.
The median age was 65 years, and 72.62% (n=61) of the study population were over 60 years old. The mean PSA concentration was 14.11 ng/ml (95% CI = [9.68–18.61]), while the median was 7.74 ng/ml. Patients living in the city of Rondonópolis accounted for 73.80% of the patients (n=62), and retirees accounted for 41.66% of the sample (n=35).
Of the 84 patients selected, 58 (69.10%) were negative for PCa. Table 1 shows the characteristics of the sample investigated, according to age, PSA value, and ISUP grade, based on positive and negative cases. Among the groups, PCa patients had a PSA level > 10 ng/ml (p=0.019). Regarding the degree of severity of PCa cases (ISUP), the highest frequency was in the intermediate category (42.3%, n=11).
Table 1. Characterization of the sample investigated, positive and negative results for PCa
|
Variables |
Results |
p-value* |
|
|
Negative PCa n=58 (69.1%) |
Positive PCa n=26 (30.9%) |
||
|
Age (years)¥ |
|||
|
43-65 |
28 (66.7) |
14 (33.3) |
0.406 |
|
66-83 |
30 (71.4) |
12 (28.6) |
|
|
PSA baseline (ng/ml)£ |
|||
|
<10 |
44 (75.9) |
13 (21.4) |
0.019 |
|
≥10 |
14 (53.8) |
13 (46.2) |
|
|
ISUPΩ Grade |
|
|
N/A |
|
Low (1) |
- |
8 (30.8) |
|
|
Intermediate (2 and 3) |
- |
11 (42.3) |
|
|
High (4 and 5) |
- |
7 (26.9) |
|
Captions: *Fisher’s Chi-Square; ¥Categorized according to median age: 65 years; £PSA: Prostate-specific antigen; ΩInternational Society of Urological Pathology.
For gene expression analysis, only one sample of a patient with negative result for PCa was excluded due to lack of sufficient material to perform the extraction step. Figure 1A shows that in the analysis of BRCA2 gene expression, there was no significant difference (p=0.786) among the medians of the groups with negative and positive results for PCa. However, as shown in Figure 1B, significantly greater expression of the BRCA1 gene (p=0.003) was detected in the group of patients who were negative for PCa.
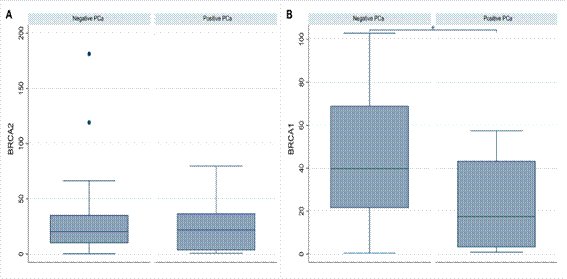
Figure 1. (A) BRCA2 gene expression among PCa and PCa-negative patients. Mann-Whitney test (p=0.786); (B) BRCA1 gene expression among PCa and PCa-negative patients. Mann-Whitney test (p=0.003)
Caption: *Indicates statistically significant difference.
To further investigate this finding, a specific gene expression analysis was performed only on fragments with negative histological results. As shown in Table 2, the median expression of the BRCA1 and BRCA2 genes was significantly higher in negative fragments of patients diagnosed with PCa when compared to fragments of patients without the neoplasia (p=0.002 and p=0.038, respectively).
Table 2. Results of BRCA1 and BRCA2 Gene Expression. Median in negative fragments of PCa cases and PCa-negative patients
|
|
Results |
|
|
|
|
Negative fragments from patients with PCa n=60 |
Negative PCa patients n=57 |
p-value* |
|
BRCA1 |
58.33 µ |
39.81 µ |
0.002 |
|
BRCA2 |
29.34 µ |
20.35 µ |
0.038 |
Captions: *Mann-Whitney Test; µMedian.
When PCa patients were stratified according to the Gleason classification, the group with a score of 7 (ISUP 2 and 3) had higher medians than did the group with a Gleason of 6 (ISUP 1) and the PCa-negative patients, as shown in Tables 3 and 4, respectively. However, no significant difference was identified among the groups.
Table 3. Results of BRCA1 and BRCA2 Gene Expression. Median according to Gleason score in PCa cases
|
|
Results |
|
|
|
|
Gleason 6 (ISUP 1) n=8 |
Gleason 7 (ISUP 2 and 3) n=11 |
p-value* |
|
BRCA1 |
15.22 µ |
39.81 µ |
0.128 |
|
BRCA2 |
18.48 µ |
35.81 µ |
0.310 |
Captions: *Mann-Whitney Test; µMedian.
When the median expression of the BRCA1 and BRCA2 genes was compared according to PSA values (up to 10 ng/ml and greater than 10 ng/ml), no difference was identified among the groups.
Table 4. Results of BRCA1 and BRCA2 Gene Expression. Median according to Gleason 7 and patients negative for PCa
|
|
Results |
|
|
|
|
Negative PCa n=57 |
Gleason 7 (ISUP 2 and 3) n=11 |
p-value* |
|
BRCA1 |
39.81 µ |
39.81 µ |
0.403 |
|
BRCA2 |
20.35 µ |
35.81 µ |
0.178 |
Captions: *Mann-Whitney Test; µMedian.
The correlation between the variables PSA, BRCA1, BRCA2 and ISUP was verified by Spearman's test which revealed that PSA was not correlated with the expression of the BRCA1 (p=0.771) or BRCA2 (p=0.435) genes. The PSA level also did not correlate with the ISUP severity measure (p=0.108).
DISCUSSION
Transrectal prostate biopsy fragments were used as biological material for the analysis of BRCA1 and BRCA2 gene expression in the present investigation. After evaluation of the pathological and immunohistochemical results, 30.9% of the patients were confirmed to have been diagnosed with PCa. This positivity rate is much greater than that presented by Segal et al.17 who performed prostate biopsies in patients with BRCA1 or BRCA2 mutations and were positive for 8.5% of the procedures.
The percentage of biopsies with positive results for PCa was very similar to that reported by Rabiau et al.18 (approximately 31%), but only 7.2% of PCa patients had a Gleason score >7. In the current investigation, 26.9% of PCa-positive samples had a Gleason score of 8, 9 or 10. These findings suggest that the requests for material collection were correctly indicated. However, this suggests a diagnosis delay since more aggressive tumors are diagnosed in more than 25% of patients.
The median age was 65 years. The mean PSA concentration was 14.11 ng/ml. These values are similar to those reported by Ibrahim et al.19 who evaluated 102 men with BRCA1 or BRCA2 mutations and found 13 PCa patients with a median age of 68 years and a mean PSA level of 14.37 ng/ml.
A comparison of the PSA values among the groups revealed that PCa patients had a PSA level > 10 ng/ml (p=0.019). In an article published by Rabiau et al.18, 98 patients who underwent prostate biopsy were evaluated, 20 of the 67 patients negative for PCa had a PSA > 10 ng/ml, whereas 54.8% of the patients with PCa had a PSA > 10 ng/ml.
There was no difference in the median BRCA2 gene expression between the groups with PCa negative and positive results. However, greater expression of the BRCA1 gene was detected in the group of patients with negative results for PCa (p=0.003). This finding is probably because other anatomopathological results, as intraepithelial neoplasia (PIN), prostatitis, and atypical proliferation of small acini (ASAP) were grouped in the PCa-negative group after immunohistochemical study reports.
Interestingly, when evaluating the expression of the BRCA1 and BRCA2 genes in negative fragments, higher medians were detected in fragments from PCa-positive patients (p=0.002 and p=0.038, respectively). This phenomenon is consistent with the concept of "field cancerization", where morphologically normal tissue already possesses molecular alterations that predispose it to malignancy. The increased expression of DNA repair genes may represent a compensatory response to a growing environment of genomic instability20. Therefore, even with a negative anatomopathological result, fragments that express these genes may indicate an unnoticed diagnosis or a case that may still evolve to PCa. This finding provides evidence for a path to be studied with regard to early diagnosis.
Rabiau et al.18 reported the overexpression of the BRCA1 gene in 26 of 57 patients with anatomopathological findings other than PCa, including NIP (Non-Ischemic Priapism) and negative results for malignancy. In contrast, in the group positive for prostate adenocarcinoma, this finding was found in 38.7% of the individuals.
When PCa patients were stratified according to Gleason classification, group 7 (ISUP 2 and 3) had greater median expression of the BRCA1 and BRCA2 genes than did group 6 (ISUP 1) and PCa-negative patients. This finding suggested that a greater expression of these genes may be related to a greater severity of the disease. However, no significant difference was identified between the groups, possibly due to the small number of patients because the collection period coincided with the beginning of the COVID-19 pandemic.
In a cohort study published by Amsi et al.21, 188 patients with PCa were evaluated. Of these, 26.1% had BRCA1 expression, 22.9% had BRCA2 expression, and 7.4% had both gene expression. A Gleason score >7 was associated with BRCA1 and BRCA2 expression (p=0.013 and p=0.041, respectively).
Ibrahim et al.18 evaluated 102 men with deleterious BRCA1 and BRCA2 mutations. PCa was the most commonly diagnosed tumor and was found in 13 patients, 11 of whom were BRCA2 positive. These tumors had higher Gleason scores and elevated PSA levels.
Stopsack et al.8 conducted a study with 575 patients with nonmetastatic PCa evaluating the association between BRCA1 expression and the development of lethal disease (cancer-specific metastasis or death) during long-term follow-up. Tumors with Gleason scores of 8–10 were more likely to be positive for BRCA1 than those with Gleason scores < 7.
Recently, Evans et al.22, evaluating 450 PCa patients in the United Kingdom, reported BRCA2 as the predominant gene in progression to metastatic disease (approximately 10% of metastatic cases). In contrast, mutations in BRCA1 were infrequent.
In an Italian study by Vatrano et al.23, 52 patients with high-risk PCa (family history of PCa, Gleason ≥ 8, and/or metastatic PCa) underwent evaluation for mutations in the BRCA1 and BRCA2 genes. Of these, 19.2% had somatic (5.7%) or germline (13.5%) mutations in BRCA, mainly in BRCA2.
The PSA value was not correlated with the expression of the BRCA1 (p=0.771) or BRCA2 (p=0.435) genes. This finding diverges from that of Oh et al.9 who evaluated 68 radical prostatectomy specimens. BRCA1-associated protein 1 expression was correlated with a high preoperative PSA level (p=0.014). However, Chen et al.24, who conducted a retrospective study with 172 individuals with PCa, found no significant difference in PSA values (p=0.623) or Gleason scores (p=0.362) among patients with and without BRCA1 and BRCA2 mutations.
This study has limitations because it only evaluated the expression of the BRCA1 and BRCA2 genes, disregarding the presence of mutations. The anatomopathological findings, as NIP, ASAP, and prostatitis were grouped into a single group that was negative for prostate cancer. The higher overall BRCA1 gene expression in patients with negative results for PCa is likely because the PCa-negative group was heterogeneous. The fragments subjected to molecular analysis were not the same as those used in the histological evaluation. The median expression was evaluated but the expression ranges within the groups were not considered. In addition, the sample size was reduced because sample collection coincided with the beginning of the COVID-19 pandemic, which limited patients' access to oncology services.
CONCLUSION
Although the expression of the BRCA1 and BRCA2 genes in transrectal prostate biopsy fragments does not seem to be related to PCa severity, the increased expression of these genes in negative fragments should be considered a warning of a disease that is undiagnosed or for a possible case in early development. Therefore, further studies should be conducted to maintain the long-term follow-up of these patients.
ACKNOWLEDGEMENTS
To “Programa de Pós-graduação em Biociências e Saúde da Universidade Federal de Rondonópolis”.
CONTRIBUTIONS
All the authors contributed to the conception and design of the study, acquisition, analysis and interpretation of the data and critical review. They approved the final version for publication.
DECLARATION OF CONFLICT OF INTEREST
There is no conflict of interests to declare.
DATA AVAILABILITY STATEMENT
Given ethical and confidentiality issues, data can be requested to the corresponding author with reasonable justification.
FUNDING SOURCES
“Fundação de Amparo à Pesquisa do Estado de Mato Grosso” (FAPEMAT), registration number 38078.544.21633.23092016.
REFERENCES
1. Instituto Nacional de Câncer. Estimativa 2023: incidência de câncer no Brasil [Internet]. Rio de Janeiro: INCA; 2022 [acesso 2023 out 24]. 160 p. Disponível em: https://www.inca.gov.br/sites/ufu.sti.inca.local/files//media/document//estimativa-2023.pdf
2. Taitt HE. Global trends and prostate cancer: a review of incidence, detection, and mortality as influenced by race, ethnicity, and geographic location. Am J Mens Health. 2018;12(6):1807-23.
3. Ferlay J, Colombet M, Soerjomataram I, et al. Cancer statistics for the year 2020: an overview. Int J Cancer. 2021;149(4):778-89. doi: https://doi.org/10.1002/ijc.33588
4. Freitas CSM, Soares AN. Efficacy of leuprorelide acetate (Eligard®) in daily practice in Brazil: a retrospective study with depot formulations in patients with prostate cancer. Int braz j urol. 2020;46:383-9. doi: https://www.doi.org/10.1590/S1677-5538.IBJU.2019.0212
5. Nicolosi P, Ledet E, Yang S, et al. Prevalence of germline variants in prostate cancer and implications for current genetic testing guidelines. JAMA Oncol. 2019;5(4):523-8. doi: https://doi.org/10.1001/jamaoncol.2018.6760
6. Kuchenbaecker KB, Hopper JL, Barnes DR, et al. Risks of breast, ovarian, and contralateral breast cancer for BRCA1 and BRCA2 mutation carriers. JAMA. 2017;317(23):2402-16. doi: https://doi.org/10.1001/jama.2017.7112
7. Nyberg T, Frost D, Barrowdale D, et al. Riscos de câncer de próstata em homens com BRCA1e portadores de mutação BRCA2: um estudo de coorte prospectivo. Eur Urol. 2020;77(1):24-35. doi: https://doi.org/10.1016/j.eururo.2019.08.025
8. Stopsack KH, Gerke T, Zareba P, et al. Tumor protein expression of the DNA repair gene BRCA1 and lethal prostate cancer. Carcinogenesis. 2020;41(7):904-8. doi: https://doi.org/10.1093/carcin/bgaa061
9. Oh H, Lee YJ, Kang SG, et al. BRCA1-associated protein 1 expression and prognostic role in prostate adenocarcinoma. Investig Clin Urol. 2020;61(2):166-72. doi: https://doi.org/10.4111/icu.2020.61.2.166
10. Patel VL, Busch EL, Friebel TM, et al. Association of genomic domains in BRCA1 and BRCA2 with prostate cancer risk and aggressiveness. Cancer Res. 2020;80(3):624-38.
11. Song WH, Kim SH, Joung JY, et al. Prostate cancer in a patient with a family history of brca mutation: a case report and literature review. J Korean Med Sci. 2017;32(2):377-81.
12. Schayek H, Haugk K, Sun S, et al. Tumor suppressor BRCA1 is expressed in prostate cancer and controls insulin-like growth factor i receptor (IGF-IR) gene transcription in an androgen receptor–dependent manner. Clin Cancer Res. 2009;15(5):1558-65.
13. Livak KJ, Schmittgen TD. Análise de dados de expressão gênica relativa usando PCR quantitativo em tempo real e o método 2 −ΔΔ CT. Methods. 2001;25(4):402-8.
14. StataR [Internet]. Versão 16.0. Lakeway: StataCorp LLC; 1996-2024c. [acesso 2025 jul 20]. Disponível em: https://www.stata.com/
15. Conselho Nacional de Saúde (BR). Resolução n° 466, de 12 de dezembro de 2012. Aprova as diretrizes e normas regulamentadoras de pesquisas envolvendo seres humanos. Diário Oficial da União, Brasília, DF. 2013 jun 13; Seção I:59.
16. Conselho Nacional de Saúde (BR). Resolução n° 510, de 7 de abril de 2016. Dispõe sobre as normas aplicáveis a pesquisas em Ciências Humanas e Sociais cujos procedimentos metodológicos envolvam a utilização de dados diretamente obtidos com os participantes ou de informações identificáveis ou que possam acarretar riscos maiores do que os existentes na vida cotidiana, na forma definida nesta Resolução [Internet]. Diário Oficial da União, Brasília, DF. 2016 maio 24 [acesso 2024 abr 7]; Seção 1:44. Disponível em: http://bvsms.saude.gov.br/bvs/saudelegis/cns/2016/res0510_07_04_2016.html
17. Segal N, Ber Y, Benjaminov O, et al. Imaging-based prostate cancer screening among BRCA mutation carriers-results from the first round of screening. Annals of Oncology. 2020;31(11):1545-52.
18. Rabiau N, Déchelotte P, Adjakly M, et al. BRCA1, BRCA2, AR and IGF-I expression in prostate cancer: correlation between RT-qPCR and immunohistochemical detection. Oncology Reports. 2011;26(3):695-702.
19. Ibrahim M, Yadav S, Ogunleye F, et al. Male BRCA mutation carriers: clinical characteristics and cancer spectrum. BMC Cancer. 2018;18(1):179.
20. Amirrad F, Pytak P, Sadeghiani‑Pelar N, et al. Prostate field cancerization and exosomes: association between CD9, early growth response 1 and fatty acid synthase. Int J Oncol [Internet]. 2020 [acesso 2025 out 11]; Disponível em: http://www.spandidos-publications.com/10.3892/ijo.2020.4980
21. Amsi PT, Yahaya JJ, Kalungi S, et al. Immunohistochemical expression of BRCA1 and BRCA2 in a cohort of Ugandan men with prostate cancer: an analytical cross-sectional study. Afr J Urol. 2020;26(1):71.
22. Evans DG, Burghel G, Schlecht H, et al. UK-based clinical testing programme for somatic and germline BRCA1/2, ATM and CDK12 mutations in prostate cancer: first results. bmjonc [Internet]. 2025 [acesso 2025 out 11];4(1). Disponível em: https://bmjoncology.bmj.com/content/4/1/e000592
23. Vatrano S, Pepe P, Pepe L, et al. BRCA mutations and prostate cancer: should urologist improve daily clinical practice? Arch Ital Urol Androl. 2025;97(2):13635. doi: https://doi.org/10.4081/aiua.2025.13635
24. Chen W, Xia W, Xue S, et al. Analysis of BRCA germline mutations in chinese prostate cancer patients. Front Oncol. 2022;12(746102):1-9. doi: https://doi.org/10.3389/fonc.2022.746102
Recebido em 5/9/2025
Aprovado em 13/10/2025
Associate-editor: Tatiana de Almeida Simão. Orcid iD: https://orcid.org/0000-0001-8509-2247
Scientific-editor: Anke Bergmann. Orcid iD: https://orcid.org/0000-0002-1972-8777
![]()
Este é um artigo publicado em acesso aberto (Open Access) sob a licença Creative Commons Attribution, que permite uso, distribuição e reprodução em qualquer meio, sem restrições, desde que o trabalho original seja corretamente citado.